FDA is Recommending Pausing the Use of Johnson and Johnson Vaccine
CDC & FDA Recommends Pausing Johnson & Johnson Vaccine
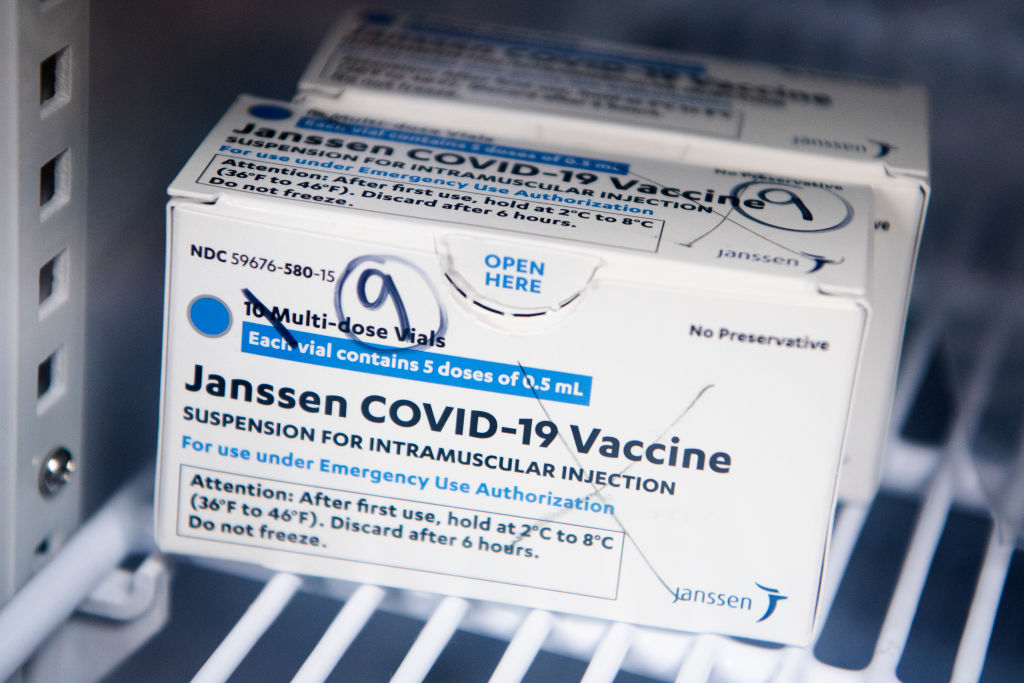
Source: Tom Williams / Getty
The FDA has announced the distribution of the Johnson & Johnson COVID-19 vaccine will be paused after six women in the U.S. had rare blog-clotting after receiving the vaccine. The women ranged in ages from 18 to 48, and while six people may not seem like a lot precautions are being taken swiftly. There have been approximately 6.8 million shots given with the majority of people not having issues.
The pausing of the Johnson & Johnson vaccine does not affect the other two vaccines from Pfizer and Moderna. But if you did receive the Johnson & Johnson shot and are having to severe stomach pain, leg cramps, or severe headaches you should call your healthcare provider right away. If you do not know what shot you received, check your CDC vaccination card where it is noted what brand shot you received. If you have an appointment scheduled to receive the Johnson & Johnson COVID-19 vaccine, it is advised to call your provider for further directions.
Get Breaking News & Exclusive Contest in Your Inbox:
[ione_media_gallery id=”3917798″ src=”https://newsone.com/” overlay=”true”]
The Latest:
- Queen Latifah’s Royal Style Moments Have Us Bowing Down Every Time
- ‘Wicked’ Costume Designer Paul Tazewell Joins The ‘Tamron Hall Show’ Designer Series As Mentor
- Are US Airport Closures Coming?
- Jay-Z To Celebrate Catalog Concert
- Sybil Wilkes Breaks Down What We Need to Know: March 18, 2026
- Kenyon Martin Presses 'Gil's Arena' Employee For Mocking His Speech Impediment & Reactions Are Mixed
- Zendaya Just Rewore Her Most Talked About Vivienne Westwood Dress & Fans Love It
- Kehlani Announces Self-Titled Album Releasing In April
- Obama Trash Talks Anthony Edwards In 1-On-1 To Announce His Presidential Center
- Airport Shutdown Fears Grow as Meteor Explosion Shakes Ohio
CDC & FDA Recommends Pausing Johnson & Johnson Vaccine was originally published on mycolumbusmagic.com









